Overview
There is worldwide interest in developing sustainable electrochemical methods for producing fuels and other chemicals that are currently derived from coal, oil, and natural gas. We contribute to these efforts by examining fundamental chemical questions regarding the mutual interactions of electrons, molecules, and materials. Our studies employ a bottom-up experimental approach that focuses on understanding the simplest (supra)molecular structures capable of supporting certain useful emergent features, such as nanoconfined catalytic sites or electronic coupling between multiple redox centers. By studying these features in systems that straddle the boundary between molecules and materials, we aim to develop insights into the design of (nano)materials with useful conductive and catalytic properties.
Redox-Active Nanocages
We design and study molecular nanocages with redox-active components that allow us to dynamically control their interactions with guests and access redox-based reactivity that can be tuned in the confined interiors of the cages. Our efforts span several areas of research:
1. Synthetic Development. We develop robust nanocages that maintain their structural integrity upon the addition or removal of multiple electrons, allowing the cages to be studied across multiple redox states. Our efforts target both metal-linked and covalently linked nanocages.

2. Interactions with Ions. We study how interactions between nanocages and ions are altered by redox changes, focusing especially on the movement and placement of protons in the cages since protons play a critical role in many important electrochemical transformations.

3. Reactivity and Catalysis. We examine how the confined environments in nanocages can be used to tune a range of important chemical reactions, spanning from electrocatalytic CO2 reduction to radical-based organic transformations. Our studies emphasize reactions that utilize the redox activity of transition metal sites installed as components of the cages.
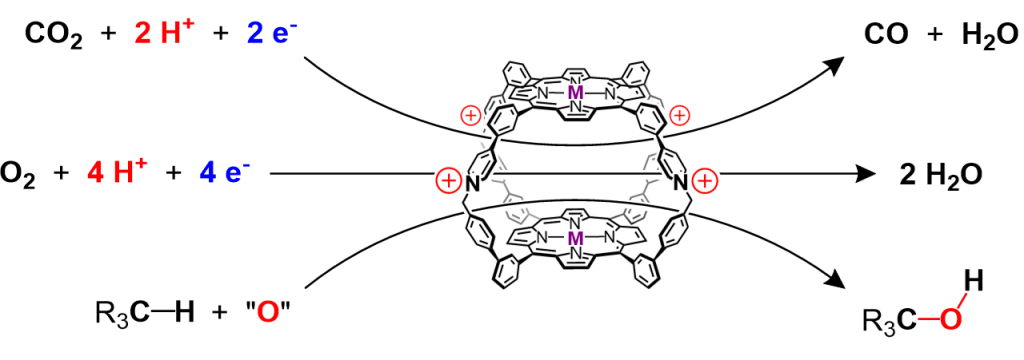
Metalloviologens
We examine the electronic and electrochemical properties of metal complexes with ligands derived from viologen cations, a storied class of redox-active organic molecules that are the basis of chemistry ranging from commercial electrochromic windows to esoteric molecular machines. Our work focuses on the ability of transition metal ions to couple the electronics of multiple viologen ligands, creating molecules in which radical electrons can delocalize across a distance of nearly 2 nm. We are interested in understanding the electronic features that give rise to this behavior, as well as in identifying emergent electrochemical properties that might result from delocalization, including expanded electrochromism, electrocatalytic activity, and solid-state conductivity.

Jump to:
Redox-Active Nanocages
Metalloviologens
